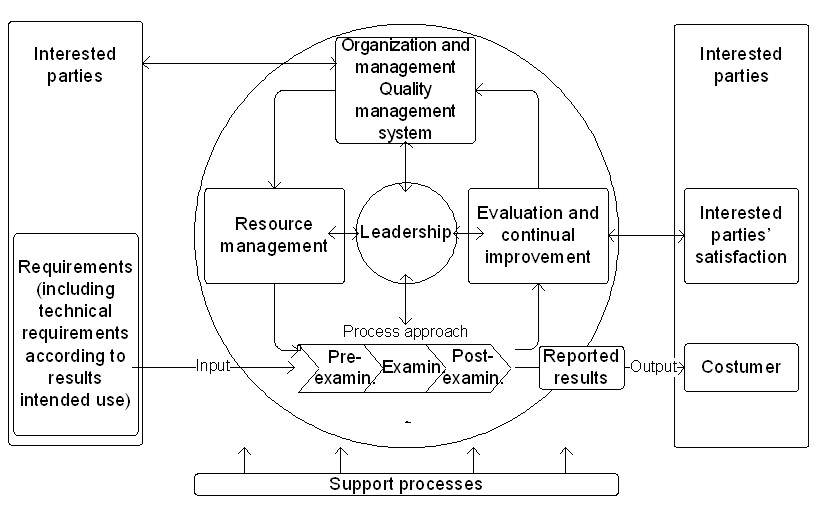
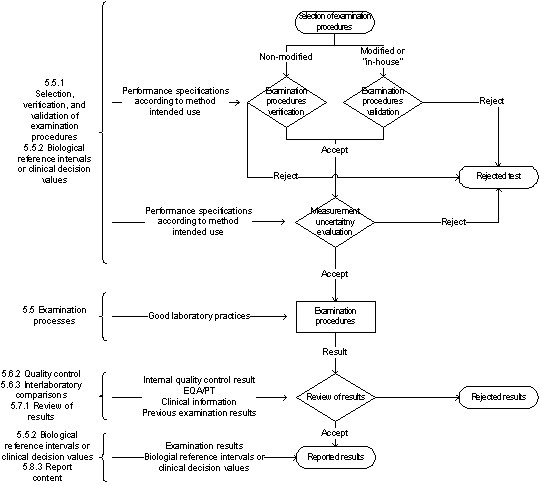
ISO 15189:2012 implementation: an update of related international standards and guidance documents for medical laboratory quality management - Document - Gale OneFile: Health and Medicine

اثر تطبيق المعيار Iso.15189 لجودة وكفاءة المختبرات الطبية في رضا المرضى في المختبرات الطبية الأردنية الخاصة | PDF | Medical Laboratory | Laboratories
UNE EN ISO 15189:2013 Medical laboratories - Requirements for quality and competence (ISO 15189:2012, Corrected
ISO 15189:2012 implementation: an update of related international standards and guidance documents for medical laboratory quality management - Document - Gale OneFile: Health and Medicine

ISO 15189:2012 Documentation Kit - Manual, Procedures in English by Global Certification Consultancy - Issuu

PDF) Integration of ISO 15189 and external quality assurance data to assist the detection of poor laboratory performance in NSW, Australia
ISO 15189:2012 implementation: an update of related international standards and guidance documents for medical laboratory quality management - Document - Gale OneFile: Health and Medicine
ISO 15189:2012 implementation: an update of related international standards and guidance documents for medical laboratory quality management - Document - Gale OneFile: Health and Medicine